TOPIC 11
Fluid and electrolytes
imbalances collaborative care and nursing management using the nursing process:
• Acid-base imbalances
• Respiratory acidosis/alkalosis
• Metabolic acidosis/alkalosis
Introduction
Acid–base
imbalance is an abnormality of the
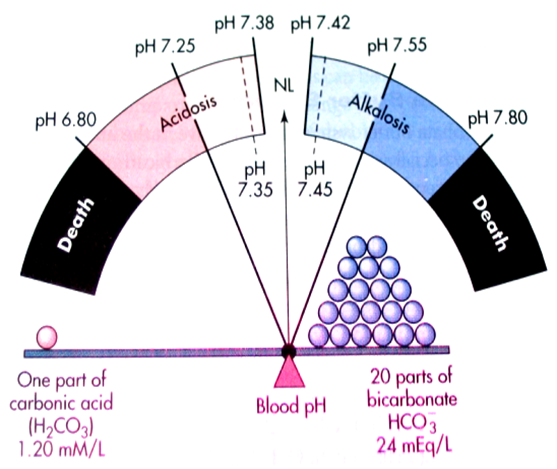
human body's normal balance of acids and bases that causes the plasma pH to
deviate out of the normal range (7.35 to 7.45).
Learning outcome
At the end of
session, participants will be able to:
1 Explain
Acid -base imbalances
2 Explain Respiratory
acidosis/alkalosis
3 Explain Metabolic acidosis/alkalosis
https:// youtu.be/VMxmDeduKR0
Facts and Definitions
1.Acid-base homeostasis is necessary to
maintain life.
2.Acid base balance must be within a
definite range for cellular function to occur.
3.The acidity of a substance, determined by
the hydrogen ion (H+) concentration; is expressed as pH.
4 Normal
function of body cells depends on regulation of hydrogen ion concentration
Hydrogen circulates throughout the body
fluids in two forms
Hydrogen circulates throughout the body
fluids in two forms:
1. The volatile H+ of carbonic acid
2. The nonvolatile form of H+ inorganic
acids (sulfuric, pyruvic, phosphoric & lactic acid)
ACID-BASE IMBALANCES
· Patients with a number of clinical conditions frequently
develop acid-base imbalances. The nurse must always consider the possibility of
acid-base imbalance in patients with serious illnesses.
· Normally the body has three mechanisms by which
it regulates acid-base balance to maintain the arterial pH between 7.35 and
7.45. These mechanisms are the buffer system, the respiratory system, and the
renal system
The buffer system is
the fastest acting system and the primary regulator of acid base balance.
The lungs help
maintain a normal pH by excreting CO2 and water, which are
by-products of cellular metabolism.
The three renal
mechanisms of acid elimination are secretion of small amounts of free hydrogen
into the renal tubule, combination of H+ with ammonia (NH3)
to form ammonium (NH4+), and excretion of weak acids.
Alterations in Acid-Base Balance
Acid-base imbalances are classified as respiratory or
metabolic. Respiratory imbalances affect carbonic acid concentrations;
metabolic imbalances affect the base bicarbonate.
· Respiratory acidosis (carbonic acid excess) occurs whenever
there is hypoventilation. Respiratory alkalosis (carbonic acid deficit) occurs whenever
there is hyperventilation.
Metabolic acidosis (base bicarbonate deficit) occurs when an
acid other than carbonic acid accumulates in the body or when bicarbonate is
lost from body fluids.
· Metabolic alkalosis (base bicarbonate excess) occurs when a
loss of acid (prolonged vomiting or gastric suction) or a gain in bicarbonate
occurs.
· Arterial blood gas (ABG) values provide valuable information
about a patient’s acid-base status, the underlying cause of the imbalance, the
body’s ability to regulate pH, and the patient’s overall oxygen status.
In cases of acid-base imbalances, the clinical manifestations
are generalized and nonspecific. The treatment is directed toward correction of
the underlying cause
Acid Base
Balance:
RATE AT WHICH RATE AT WHICH
ACIDS / BASES = ACIDS
/ BASES
ARE PRODUCED ARE EXCRETED
This balance
results in a stable concentration of Hydrogen ions (H+) and it is
this
concentration that is called pH
pH value: Normal
value is 7 < 7: Acidic
>
7: Alkaline
Arterial
Blood: 7.35 – 7.45
At pH 7.35 – 7.45:
Cell
wall /membrane integrity is maintained
Speed
of cellular enzymatic actions is maintained
Acid-Base Regulation
through the:
1.
Chemical buffering system
2.
Biological buffering system
3.
Physiological buffering system
THESE BUFFER SYSTEMS ABSORB OR
RELEASE H+ IONS TO CORRECT
ACID BASE BALANCE
1. Chemical Buffering System
This system is in the ECF It is called Carbonic Acid-Bicarbonate Buffer System .This system is the first to react to pH changes in the ECF. This System reacts within seconds
cellular metabolism--CO2 is produce ------
As CO2
is produced, H+ ions increased If CO2
not removed by the Lungs,the following
takes place
CO2
+ H2O ............ H2CO3 ........ H+ + HCO3
Carbon
+ Water Carbonic
Acid Hydrogen+bicarbonate dioxide ion
Carbonic Acid -Bicarbonate Buffer System
This is the largest buffer system in the Extracellular Fluid
PHYSIOLOGICAL BUFFERING SYSTEM
There are 2 physiological
buffering systems: LUNGS
KIDNEYS
3.1 Lungs: Act rapidly to an Acid-Base
Imbalance (before the biological system)
When concentration of
H+ alter, the rate and depth of respiration will
alter:Eg:
Metabolic Acidosis Rate of respiration is increased More CO2 exhaled
<7 pH returns to 7 acidity corrected
Metabolic Alkalosis Rate of respiration is decreased CO2 retained
pH returns to 7 Alkalinity corrected
3.2 Kidneys: Kidneys
take from a few hours to several days to regulate acid-base balance
When pH <7 Kidneys reabsorb HCO3 pH
is corrected
When pH >7 Kidneys excrete HCO3 pH is
corrected
When H+> Kidneys excrete H+
ions by:
a) combining H+
and PO43- to form Phosphoric Acid H3PO4
Phosphoric acid is then excreted in the urine
b) Amino acid is changed to Ammonia
in the renal tubules
Ammonia combines with H to form
Ammonium Ammonium is then
excreted in the urine
DISTURBANCES IN ACID BASE BALANCE
Checking Acid-Base Balance in a Client:
Arterial Blood Gas Analysis (ABG)
Parameters of Measurement in ABG: 1. pH
2.
PaCO2
3.
PaO2
4.
O2 Saturation
5.
Base Excess
6.
HCO3
pH: Measures
H+ concentration in the body fluids
PaCO2 Partial pressure of CO2 in the artery (reflects
depth of pulmonary ventilation)
Normal
value: 35-45mm Hg
<
35-45 mm Hg Hyperventilation has
occurred
> 45 mm Hg Hypoventilation has occurred
PaO2 Partial
pressure of O2 in arterial blood
Normal
value: 80-100mm Hg
<
60mm Hg Lead to anaerobic
metabolism > Lactic
acid production > Metabolic
Acidosis
> 100mm Hg Hypoxemia
Oxygen Saturation: Arterial Hemoglobin saturated with
O2
Normal Value: 95% - 100%
HCO3: Excreted and reabsorbed by the Kidneys to maintain Acid-Base
Balance
Principle
Buffer in the ECF
Normal
Value: 22 – 26
mEq/L
<
than 22-26mEq/L Metabolic
Acidosis
> than 22-26mEq/l Metabolic Alkalosis
TYPES OF ACID-BASE IMBALANCES:
1.
Respiratory Acidosis: Carbonic Acid Excess
2.
Respiratory Alkalosis : Carbonic Acid Deficit
3. Metabolic Acidosis: Bicarbonate Deficit
4. Metabolic Alkalosis : Bicarbonate Excess
PaCO2
• Carbonic acid level is
measured by PaCO2 value of the blood
• partial pressure of
CO2 in the arterial blood
• normal value: 35 – 45
mm Hg
Bicarbonate
( HCO3)
• The most abundant base
in the body fluids
• Metabolic end product
of fats and carbohydrates
• Normal serum value 22
– 26 mmHg
ACID - BASE IMBALANCE
Classifications
1. Acidosis
or alkalosis
a. Acidosis: Hydrogen ion
concentration in blood increases above
normal and pH is below 7.35
b. Alkalosis: Hydrogen ion concentration
in blood decreases below normal and pH is above 7.45
Analysis
of Arterial Blood Gases
Step1: Classify the
Arterial Gas
Normal: 7.35 – 7.45
Acidosis: below
7.35
Alkalosis: above
7.45
Step 2: Assess PaCO2
Normal: 35 – 45
mmHg
Respiratory
Acidosis: above 45 mmHg
Respiratory
Alkalosis: below 35 mmHg
Step 3: Assess HCO3
Normal: 22 –26
mEq/L
Metabolic
Acidosis: below 22 mEq/L
Metabolic Alkalosis:
above 26 mEq/L
Step 4 : classify
degree of compensation
pH
is normal: fully compensated
pH is not normal: partially compensated
Respiratory acidosis
Respiratory
acidosis is a condition that occurs when your lungs can’t remove all of the
carbon dioxide produced by your body. This causes the blood and other body
fluids to become too acidic.
•
pH < 7.35
•
pCO2 > 45 mm Hg (excess carbon dioxide
in the blood)
•
Respiratory system impaired and retaining CO2
causing acidosis
Causes of respiratory acidosis
·
Airway diseases, such as asthma and COPD
·
Lung tissue diseases, such as pulmonary fibrosis, which causes scarring and
thickening of the lungs
·
Diseases that can affect the chest,
such as scoliosis
·
Diseases that affect the nerves and
muscles that signal the lungs to inflate or deflate
·
Medicines that suppress breathing,
including narcotics (opioids), and "downers," such as
benzodiazepines, often when combined with each other or alcohol
·
Severe obesity, which restricts how
much the lungs can expand
·
Obstructive sleep apnea
·
Chest
deformities, such as kyphosis ,
·
Chest injuries, Chest
muscle weakness, Long-term (chronic) lung disease
·
Neuromuscular
disorders, such as myasthenia gravis, muscular dystrophy
- Overuse of
sedative drugs, causing decreased respiration
a Acute respiratory failure from airway obstruction
b.
Over-sedation from anesthesia or narcotics
c.
Some neuromuscular diseases that affect ability to use chest muscles
d. Chronic respiratory problems, such as
Chronic Obstructive Lung Disease
Signs and
Symptoms of respiratory acidosis
a. Compensation s/s: kidneys
respond by generating and reabsorbing
bicarbonate ions,
so HCO3 >26 mm
Hg
b. Respiratory: hypoventilation,
slow or shallow respirations
c. Neuro: headache, blurred
vision, irritability, confusion – cerebral vessels dilate
d. Respiratory collapse leads to
unconsciousness and
cardiovascular collapse
e Confusion, Fatigue,
Lethargy, Shortness of breath, Sleepiness
Management respiratory acidosis
Therapeutic
measures that may be lifesaving in severe hypercapnia and respiratory acidosis
include endotracheal intubation with mechanical ventilation and noninvasive
positive pressure ventilation (NIPPV) techniques such as nasal continuous
positive-pressure ventilation (NCPAP) and nasal bilevel ventilation.
Early
recognition of respiratory status and treat cause
B. Restore
ventilation and gas exchange
-CPR for
respiratory failure with oxygen
supplementation
-intubation
and ventilator support if indicated
C. Treatment
of respiratory infections
-bronchodilators;
antibiotic therapy
D. Reverse
excess anesthetics and narcotics
-
naloxone (Narcan)
E Continue respiratory assessments-
monitor arterial blood gas (ABG) results
Chronic respiratory conditions:
Breathe in response to low oxygen levels –
Adjusted to
high carbon dioxide level through metabolic compensation (therefore,
high CO2 would not trigger breathing)
-Treat with
no higher than 2 liters O2 per cannula (carbon dioxide narcosis)
Drug treats respiratory
acidosis
Respiratory
Acidosis Medication: Beta2 Agonists, Anticholinergics, Respiratory, Xanthine
Derivatives, Corticosteroids, Benzodiazepine Toxicity Antidotes, Opioid
Antagonist
Interpret
these ABG results
pH - 7.25 - acidic
PaCO2 – 50mmHg - acidic
HCO3 – 22 mEq/L – normal
- Respiratory acidosis, uncompensated
pH - 7.37 - normal
PaCO2 - 60 mmHg- acidic
HCO3 - 38 mEq/L - alkaline
- respiratory acidosis, fully compensated with
metabolic alkalosis
Acidosis
https://youtu.be/-4HwKsDgf7Y
Respiratory alkalosis
Respiratory
alkalosis is a condition marked by a low level of carbon dioxide in the blood
due to breathing excessively
- ppH >7.45
- CO2 < 35 mm Hg
- Carbon dioxide deficit, secondary to hyperventilation
Respiratory alkalosis may be produced
as a result of the following causes:
Hyperventilation with anxiety from:
- uncontrolled fear,
pain, stress (e.g. women in labor, trauma victims)
-High fever. Stress[1]
Mechanical ventilation
during anesthesia
·
Pulmonary disorder[3]
·
Thermal insult[6]
·
High altitude areas[7]
·
Salicylate poisoning (aspirin overdose)[7]
·
Hyperventilation (due
to heart disorder or other, including improper mechanical ventilation)[1][8]
·
Vocal cord paralysis (compensation
for loss of vocal volume results in over-breathing/breathlessness).[9]
·
Liver disease[7]
·
drugs cause respiratory alkalosis
·
Iatrogenic
causes of respiratory alkalosis include medications like progesterone,
methylxanthines (e.g., theophylline), salicylates (also cause primary metabolic
acidosis), catecholamines and nicotine as well as excessive minute
ventilation provided by mechanical ventilation (especially in chronic
obstructive pulmonary
The diagnosis
of respiratory alkalosis is done via
test that measure the oxygen and carbon dioxide levels (in the blood), chest x-ray
and a pulmonary function test ,
There are two types of respiratory
alkalosis: chronic and acute as a
result of the 3–5 day delay in kidney compensation of the abnormality.[13][3]
·
Acute
respiratory alkalosis occurs rapidly, have a high pH
because the response of the kidneys is slow.[14]
·
Chronic
respiratory alkalosis is a more long-standing
condition, here one finds the kidneys have time to decrease the bicarbonate
level.[14]
Signs and Symptoms:
Compensation: Kidneys-eliminating bicarbonate ions
HCO3<22mmHg
Respiratory-hyperventilating :shallow,rapid breathing
Neuro-panicked,light-headed.tremors-may develop tetany.numb hands and feet (carpopedal) r/t symptoms of hypocalcemia
(elevated pH more
Ca ions are bound to serum albumin and less ionized “active” calcium available
for nerve and muscle conduction)
•
seizures,
loss of consciousness - due to cerebral constriction
•
Cardiac:
(H+ pulled from the cells in exchange of K+ -hypokalemia)
- palpitations,
sensation of chest tightness
- ECG changes
Management of
Respiratory Alkalosis
Encourage client to breathe slowly in a paper bag
to rebreathe CO2
* Breathe with the patient
* Provide emotional support and
reassurance
* Anti-anxiety agents
* Sedation
•
On
ventilator, adjustment of ventilation settings (decrease rate and tidal volume)
•
Prevention
- pre-procedure
teaching
- emotional support
-
monitor blood gases
Metabolic Acidosis
https://youtu.be/0Er2a9-OxGc
Deficit of bicarbonate in the
blood
•
pH <7.35
•
NaHCO3 <22 mEq/L
Causes of
Metabolic Acidosis
•
Caused by an excess of acid, or loss of bicarbonate
from the body
•
Acute lactic acidosis from tissue hypoxia:
(lactic acid produced from anaerobic metabolism
with shock, cardiac arrest)
•
Ketoacidosis:
(fatty acids are released and converted to
ketones when fat is used to supply glucose needs
as in uncontrolled Type 1 diabetes or starvation)
•
Acute or chronic renal failure
(kidneys unable to regulate electrolytes)
•
Excessive bicarbonate loss (severe
diarrhea, intestinal suction, bowel fistulas)
•
results from other disease and often accompanied by
electrolyte and fluid imbalances
•
Hyperkalemia often occurs as hydrogen ions enter cells
to lower pH and K+ enter intravascular
Signs and
Symptoms
•
Compensation s/s:
-
increase the depth and rate of respiration (an effort to lower the CO2
in the blood paCO2
<35 mm Hg)
•
Neuro changes: (electrolytes imbalance)
- headache, weakness,
fatigue, confusion, stupor and coma
•
Cardiac: dysrhythmias and possibly cardiac arrest from
hyperkalemia
•
GI: anorexia, nausea, vomiting
·
Hyperventilating., Shortness of
breath.
·
Fatigue., Chronic exhaustion.
Headaches., Drowsiness. , Confusion. Dizziness
·
Sweating, Breathlessness.
·
Numbness and /or tingling in your
fingertips, toes and lips.
·
Irritability. , Nausea. , Muscle
spasms or twitching
Nursing Diagnoses:
a. Decreased Cardiac Output
b. Risk for Excess Fluid Volume
c. Risk for Injury
Metabolic Acidosis
https://youtu.be/0Er2a9-OxGc
Deficit of bicarbonate in the
blood
•
pH <7.35
•
NaHCO3 <22 mEq/L
Causes of
Metabolic Acidosis
•
Caused by an excess of acid, or loss of bicarbonate
from the body
•
Acute lactic acidosis from tissue hypoxia:
(lactic acid produced from anaerobic metabolism
with shock, cardiac arrest)
•
Ketoacidosis:
(fatty acids are released and converted to
ketones when fat is used to supply glucose needs
as in uncontrolled Type 1 diabetes or starvation)
•
Acute or chronic renal failure
(kidneys unable to regulate electrolytes)
•
Excessive bicarbonate loss (severe
diarrhea, intestinal suction, bowel fistulas)
•
results from other disease and often accompanied by
electrolyte and fluid imbalances
•
Hyperkalemia often occurs as hydrogen ions enter cells
to lower pH and K+ enter intravascular
Signs and
Symptoms
•
Compensation s/s:
-
increase the depth and rate of respiration (an effort to lower the CO2
in the blood paCO2
<35 mm Hg)
•
Neuro changes: (electrolytes imbalance)
- headache, weakness,
fatigue, confusion, stupor and coma
•
Cardiac: dysrhythmias and possibly cardiac arrest from
hyperkalemia
•
GI: anorexia, nausea, vomiting
·
Hyperventilating., Shortness of
breath.
·
Fatigue., Chronic exhaustion.
Headaches., Drowsiness. , Confusion. Dizziness
·
Sweating, Breathlessness.
·
Numbness and /or tingling in your
fingertips, toes and lips.
·
Irritability. , Nausea. , Muscle
spasms or twitching.
Nursing Diagnoses:
a. Decreased Cardiac Output
b. Risk for Excess Fluid Volume
c. Risk for Injury
Metabolic acidosis develops when too
much acid is produced in the body. It can also occur when the kidneys cannot remove
enough acid from the body. There are several types of metabolic acidosis:
·
Diabetic acidosis (also called
diabetic ketoacidosis and DKA) develops when substances called ketone bodies
(which are acidic) build up during uncontrolled diabetes.
·
Hyperchloremic
acidosis is caused by the loss of too much sodium bicarbonate from the body,
which can happen with severe diarrhea.
·
Kidney
disease (uremia, distal renal tubular
acidosis or proximal renal
tubular acidosis).
·
Lactic
acidosis.
·
Poisoning
by aspirin, ethylene glycol (found in antifreeze), or methanol.
·
Severe dehydration.
Lactic acidosis is a buildup
of lactic acid. Lactic acid is
mainly produced in muscle cells and red blood cells. It forms when the body
breaks down carbohydrates to use for energy when oxygen levels are low.
·
Cancer
·
Carbon monoxide
poisoning
·
Drinking
too much alcohol
·
Exercising
vigorously for a very long time
·
Liver
failure
·
Low
blood sugar (hypoglycemia)
·
Medicines,
such as salicylates, metformin, anti-retrovirals
·
MELAS
(a very rare genetic mitochondrial disorder that affects energy production)
·
Prolonged
lack of oxygen from shock, heart failure, or severe anaemia
·
Seizures
·
Sepsis -- severe
illness due to infection with bacteria or other germs
·
Severe
asthma
Metabolic
Alkalosis
•
pH >7.45
•
HCO3
> 26 mEq/L
Causes of
Metabolic Alkalosis
Caused by a
bicarbonate excess, due to loss of acid, or a bicarbonate excess in the body
• 1 Loss of
hydrogen and chloride ions through excessive vomiting, gastric suctioning, or
excessive diuretic therapy
•
2. Response to hypokalemia
•
3. Excess ingestion of bicarbonate rich
antacids or excessive treatment of acidosis with Sodium Bicarbonate
• The most common causes are volume
depletion (particularly when involving loss of gastric acid and
chloride (Cl) due to recurrent vomiting or nasogastric suction) and diuretic
use. Metabolic alkalosis involving loss or excess secretion of Cl is termed
chloride-responsive
Signs and
symptoms
1
Compensation: Lungs respond by decrease
the depth and rate of respiration in effort to retainCO2 and increase pH
2. Neuro:
altered mental status, numbness and tingling around mouth, fingers, toes, dizziness, muscle
spasms (similar to hypocalcemia due to less ionized calcium levels)
3.
Hypokalemia - H+ moves out of cell & K+ moves inside cell
Metabolic Alkalosis Treatment & Management
The management of metabolic alkalosis depends
primarily on the underlying etiology and on the patient’s volume status. In the
case of vomiting, administer antiemetics, if possible. If continuous gastric
suction is necessary, gastric acid secretion can be reduced with H2-blockers or
more efficiently with proton pump inhibitors. In patients who are o
on thiazide or loop diuretics, the dose
can be reduced or the drug can be stopped if appropriate. Alternatively, a
potassium-sparing diuretic or acetazolamide can be added.
1 Correcting underlying cause will often improve alkalosis
2.Restore fluid volume and correct
electrolyte imbalances (usually IV NaCl with KCL).
3.With severe cases, acidifying solution may
be administered.
Restore fluid volume and correct electrolyte
imbalances (usually IV NaCl with KCL) With severe cases, acidifying solution may be administered
Metabolic alkalosis
is corrected with the aldosterone antagonist spironolactone or with
other potassium-sparing diuretics (eg, amiloride, triamterene). If the
cause of primary hyperaldosteronism is an adrenal adenoma or carcinoma,
surgical removal of the tumor should correct the alkalosis.
Correcting underlying cause will often improve alkalosis
Nursing Diagnoses:
a. Impaired
Gas Exchange
b. Ineffective Airway Clearance
c. Risk for Injury
Interpreting
results
•
pH - 7.5
•
PaCO2 - 40 mmHg
HCO3 -
30 mEq
•
pH - 7.5-
alkalosis
•
PaCO2 - 40 mmHg - normal
•
HCO3 - 17 mEq/L - metab alkalosis
•
Metabolic
alkalosis ,uncompensated
Metabolic Acidosis
Metabolic acidosis occurs when your body produces too much acid ,or your kidney don't reproduces
What causes metabolic acidosis?
The four leading causes of metabolic acidosis include:
Diabetes-related acidosis.
Diabetes-related acidosis develops when ketone bodies build up in your body
from untreated diabetes. Your body produces ketone bodies while it turns
(metabolizes) fats into energy. Your body uses ketone bodies for energy when
sugars (glucose) aren’t available.
Hyperchloremic acidosis.
Hyperchloremic acidosis develops when your body loses too much sodium bicarbonate.
It may occur if you take too many laxatives or have severe diarrhea.
Lactic acidosis.
Lactic acidosis develops when you have too much lactic acid in your body. Lactic
acid is an organic acid that your muscle cells and red blood cells produce for
energy when you don’t have a lot of oxygen in your body. Causes include liver
failure, low blood sugar, alcohol
use disorder, cancer and intense exercise.
Renal tubular acidosis.
Renal tubular acidosis develops when your kidneys don’t pass enough acids into
your pee. As a result, your blood becomes more acidic.
· Causes include untreated diabetes, the loss of bicarbonate in
your body and kidney conditions.
Unmanageable diarrhea and kidney failure are the most common
causes of metabolic acidosis. remove enough
acids from your blood.
Common
signs and symptoms of metabolic acidosis include:
·
Accelerated heartbeat (tachycardia).
·
Confusion or dizziness.
·
Feeling very tired (fatigue).
·
Loss of appetite.
·
Headache.
·
Rapid breathing or long, deep breathing.
·
Nausea
and vomiting.
·
Feeling weak.
·
Breath that smells sweet or fruity.

View
Dear students, please complete this quiz to test your
understanding of Topic 11
Interpret these
ABG results:
pH - 7.25
PaCO2 – 50mmHg
HCO3 – 22 mEq/L
Answer
pH - 7.25 – acidic
PaCO2
– 50mmHg - acidic
HCO3 – 22 mEq/L –
normal
- Respiratory
acidosis, uncompensated
Interpret these ABG
results:
pH -
7.37
PaCO2
- 60 mmHg
HCO3
- 38 mEq/L
Answer
pH -
7.37 - normal
PaCO2
- 60 mmHg- acidic
HCO3
- 38 mEq/L - alkaline
- respiratory acidosis, fully compensated with
metabolic alkalosis
Blood and urine tests can help diagnose
Interpret these ABG
results:
pH -
7.37
PaCO2
- 60 mmHg
HCO3
- 38 mEq/L
Answer
pH -
7.37 - normal
PaCO2
- 60 mmHg- acidic
HCO3
- 38 mEq/L - alkaline
- respiratory acidosis, fully compensated with
metabolic alkalosis
Blood and urine tests can help diagnose
Treatments include:
- Sodium citrate if you have kidney disease or
kidney failure.
- Fluids delivered through a vein in your arm
(IV fluids).
- IV sodium bicarbonate, which helps balance the
acids in your blood.
- Insulin if you have diabetes-related acidosis.
- Removing toxic substances from your blood,
including aspirin, methanol (a substance in adhesives, paints and
varnishes) or ethylene glycol (a substance in antifreeze) Inotropes
help your heart beat stronger, which helps get more oxygen in your body
and lowers the amount of acids in your blood. .
Treatment plan, which may include medications and changes to
your lifestyle
- Monitoring
your blood sugar closely.
- Taking
medicines as prescribed by your healthcare provider.
- Eating
low-acid, high-alkali foods.
- Limiting
the amount of alcohol, you consume.
Prevention
You can’t prevent metabolic acidosis. However, you can help
reduce your risk
- Drinking
a lot of water and other fluids.
- Managing
your blood sugar levels if you have diabetes.
- Reducing
the amount of alcohol that you consume. Moderate alcohol consumption in
men and people assigned male at birth (AMAB) is two drinks or fewer per
day. In women and people assigned female at birth (AFAB), moderate Severe cases may involve kidney or other
organ failure and death
What is the difference between metabolic acidosis and respiratory acidosis?
Metabolic acidosis involves your digestive
system and your urinary
system. Your kidneys can’t properly filter acids from your bloodstream.
Kidney disease, kidney failure, untreated diabetes, loss of bicarbonate and
blood poisoning may cause a more acidic pH in your body.
Respiratory acidosis involves your respiratory
system. Your lungs can’t remove enough carbon dioxide from your
bloodstream. Asthma, brain injuries and excessive or disordered substance use
may affect your lungs’ ability to remove carbon dioxide. my risk of
Discussion Forum
View Make forum post 1
Students, please answer
the questions in this forum
How can I reduce my risk of developing metabolic acidosis?
What causes metabolic acidosis?
Interpret these ABG results:
•
Q1 pH - 7.5
•
PaCO2 - 40 mmHg
HCO3 - 30
mEq/L
•
pH - 7.5-
alkalosis
•
PaCO2 - 40 mmHg - normal
•
HCO3 - 17 mEq/L - metabolic alkalosis
•
Metabolic
alkalosis, uncompensated
Q2 pH - 7.38
•
PaCO2 – 50 mmHg
•
HCO3 – 35 mEq/L
Answer Q2 pH -
7.38 - normal (acidosis)
•
PaCO2 – 50 mmHg - acidosis
•
HCO3 – 28 mEq/L
- alkalosis
•
Respiratory acidosis, fully compensated with metabolic
alkalosis
•
Q 3 Ph 7.39
•
PaCO2 – 32 mmHg
HCO3 - 20
mEq/ L
•
Answer Q3 v Ph
7.39 - normal ( acidosis)
•
PaCO2 – 32 mmHg
- alkalosis
•
HCO3 - 20 mEq/ L
- acidosis
•
Metabolic acidosis, fully compensated with respiratory
alkalosis
•
Q 4 Ph 7.49
•
PCO2 – 50 mmHg
•
HCO3 – 32 mEq/L
AnswerQ4 Ph 7.49 - ( abnormal) alkalosis
•
PCO2 – 50 mmHg
- acidosis
•
HCO3 – 32 mEq/L
- alkalosis
•
Metabolic alkalosis, partially compensated with respiratory
acidosis


