Topic 3: Inorganic Compounds
3.1 Organic versus Inorganic Compound
TLO 1: Compare and contrast inorganic and organic compounds.
Most of the chemicals in your body exist in the form of compounds. Biologists and chemists divide these compounds into two principal classes: inorganic compounds and organic compounds.
- An inorganic compound is a substance that does not contain both carbon and hydrogen. A great many inorganic compounds do contain hydrogen atoms, such as water (H2O) and the hydrochloric acid (HCl) produced by your stomach. In contrast, only a handful of inorganic compounds contain carbon atoms. Carbon dioxide (CO2) is one of the few examples.
- An organic compound, then, is a
substance that contains both carbon and hydrogen. Organic compounds are
synthesized via covalent bonds within living organisms, including the
human body. Recall that carbon and hydrogen are the second and third most
abundant elements in your body. You will soon discover how these two
elements combine in the foods you eat, in the compounds that make up your
body structure, and in the chemicals that fuel your functioning.

Figure 1 Examples of organic and inorganic compounds. (source: https://sciencenotes.org/)
3.2 Properties of water
TLO 2: Identify the characteristic of water that makes it essential for life
The adult’s body are composed as much as 70% of water. Water is the most important and abundant inorganic compound in all living systems. We might be able to survive without food for a week, but without water we would die in matter of days. Nearly all the body’s chemical reactions occur in a watery medium.
3.2.1 Water as solvent
A mixture is a combination of two or more substances, each of which maintains its own chemical identity. In other words, the constituent substances are not chemically bonded into a new, larger chemical compound. The concept is easy to imagine if you think of powdery substances such as flour and sugar; when you stir them together in a bowl, they obviously do not bond to form a new compound. The room air you breathe is a gaseous mixture, containing three discrete elements—nitrogen, oxygen, and argon—and one compound, carbon dioxide. There are three types of liquid mixtures, all of which contain water as a key component. These are solutions, colloids, and suspensions.
For cells in the body to survive, they must be kept moist in a water-based liquid called a solution. In chemistry, a liquid solution consists of a solvent that dissolves a substance called a solute. An important characteristic of solutions is that they are homogeneous; that is, the solute molecules are distributed evenly throughout the solution. If you were to stir a teaspoon of salt into a glass of water, the salt would dissolve into salt molecules separated by water molecules. The ratio of salt to water in the left side of the glass would be the same as the ratio of salt to water in the right side of the glass. If you were to add more salt, the ratio of salt to water would change, but the distribution—provided you had stirred well—would still be even.
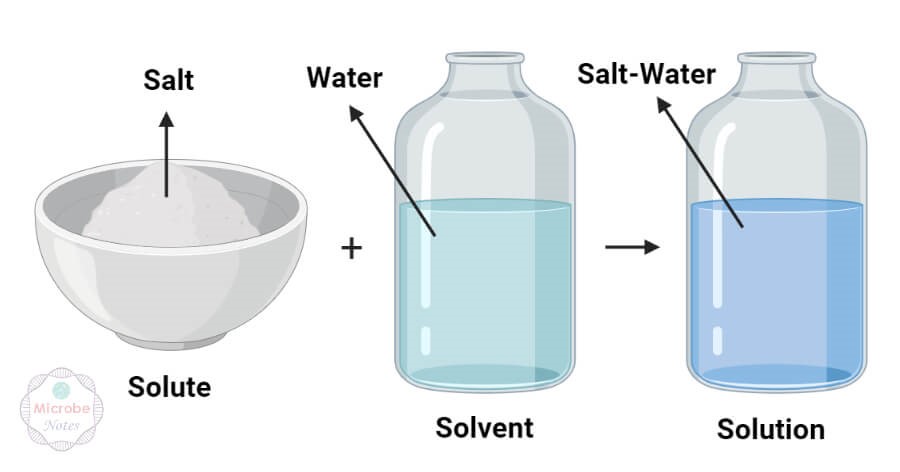
Figure 2 Solute, solvent and solution (source: https://thechemistrynotes.com/)
Water is considered the “universal solvent” and it is believed that life cannot exist without water because of this. Water is certainly the most abundant solvent in the body; essentially all of the body’s chemical reactions occur among compounds dissolved in water.
Because water molecules are polar, with regions of positive and negative electrical charge, water readily dissolves ionic compounds and polar covalent compounds. Such compounds are referred to as hydrophilic, or “water-loving.” As mentioned above, salt dissolves well in water. This is because salt molecules contain regions of hydrogen-oxygen polar bonds, making it hydrophilic. Nonpolar molecules, which do not readily dissolve in water, are called hydrophobic, or “water-fearing.”
The ability of water to form solutions is essential to health and survival. Because water can dissolve so many different substances, it is an ideal medium for metabolic reactions. Water enables dissolved reactants to collide and form products. Water also dissolves waste products, which allows them to be flushed out of the body in the urine.
3.2.2 Water in chemical reactions
Water serves as the medium for most chemical reactions in the body and participates as a reactant or product in certain reactions. During digestion, for example, decomposition reactions break down large nutrient molecules into smaller molecules by the addition of water molecules. This process is known as the hydrolysis.
Hydrolysis reactions enable dietary nutrients to be absorbed into the body. By contrast, when two smaller molecules join to form a larger molecule in a dehydration synthesis reaction (de- = from, down, or out; hydra- = water), a water molecule is one of the products formed.
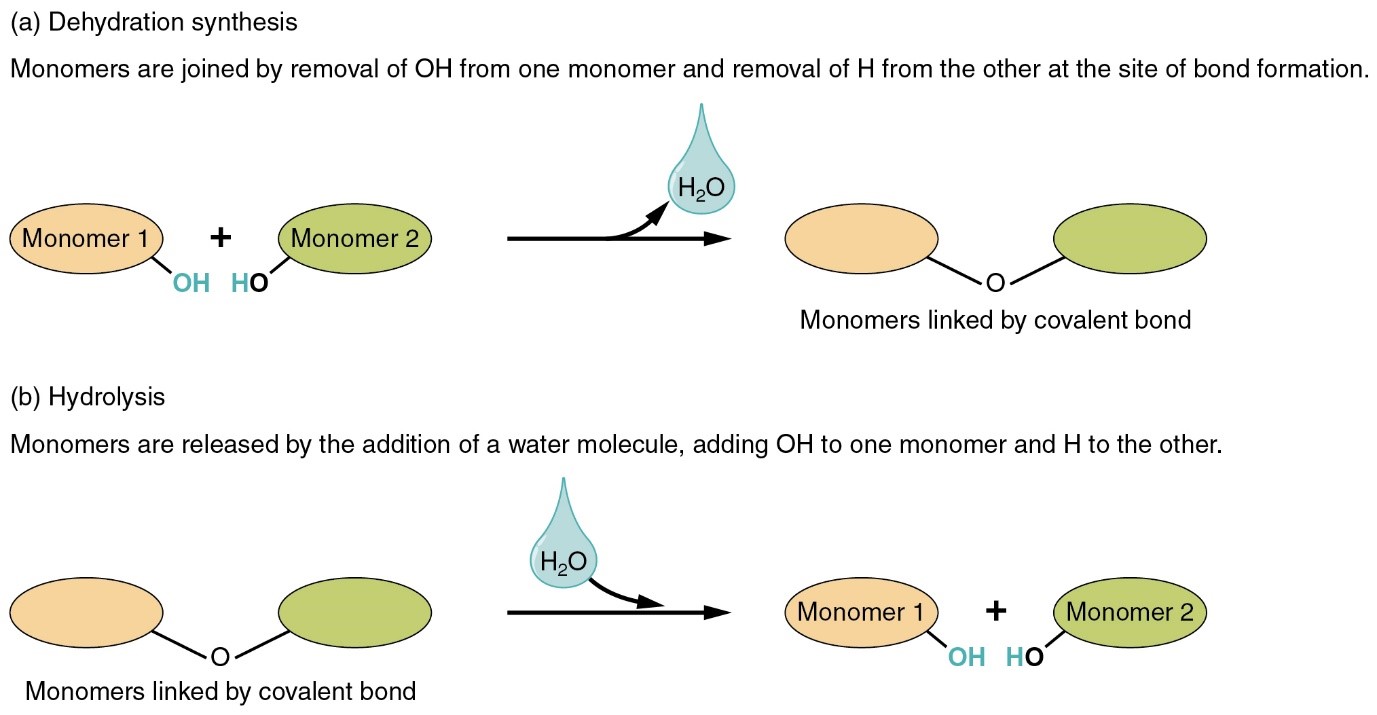
Figure 3 Dehydration and hydrolysis process that either use or produce water. (source: https://openstax.org/)
- View